Current Research
Our holistic view on catalysis
Our research is focused on the synthesis of organometallic complexes having in mind their immediate applications as catalysts to the organic synthesis. We are trying to develop efficient and low-waste catalytic methods for selective synthesis of natural and pharmaceutically relevant compounds, transformation of biomass, sustainable production of fine chemicals, etc.
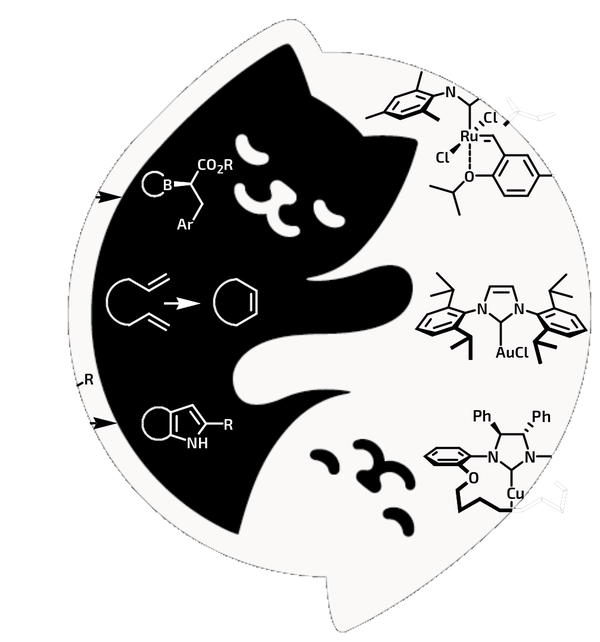
Yin and yang of Cats and Transformations
Yin-yang artwork ©2018 Irina Mir, Valdivia, Chile (instagram/irmirx/)
Our goal is to develop innovative strategies and industry-friendly protocols that allow to produce various target molecules utilising the smallest possible amount of catalyst in a clean and environment friendly ways.
New concepts in catalysts design
Concepts developed by us, such as the EWG activation / EDG deactivation of the Hoveyda-Grubbs catalyst, novel Ru···E chelates (E = S, N, Br, I), 'sticky' ammonium-tags, were not only used in Warsaw to 'produce' scientific publications, but were almost instantly assimilated by industrial players that deployed them to the market.
Using these concepts we developed a number of innovative ruthenium catalysts that have been commercialised and are available e.g. from Strem Inc. USA, or TCI Japan.
Tailored ligands for better catalysis
New specialised catalysts developed by us are based on tailored NHC and CAAC ligands. The innovative ruthenium catalysts, being recently patented, contain i.a. unsymmetrical NHC ligands and exhibit perfect selectivity in challenging ethenolysis of oleic esters from biomass, in cross-metathesis of α-olefins and in RCM of dienes leading to macrocyclic musk-smelling compounds. A copper complex bearing chiral NHC ligand showed high enantioselectivity in the β-borylation reaction.
New substrates, conditions and supportive technologies
We were lucky to introduce a number of partners for cross-metathesis reaction, such as vinyl sulfones and sulfoxides, P-chiral vinyl phosphine oxides, vinyl halides, allyl nitro compounds and more. We also developed new ring closing technique to obtain macrocyclic compounds, including macrocyclic musks, at relatively high concentration.
We have published a series of papers on the doping effect of fluorinated aromatic hydrocarbon solvents on the performance of the olefin metathesis reaction. Together with the respective technology owner, Apeiron Synthesis S.A. we worked on an innovative scavenger, that allows to obtain low levels of heavy metal contamination after a simple and waste-free purification.
Current areas of investigation encompass the design of new user-friendly, supported or 'self-cleaning' catalysts and extending their application scope utilising flow techniques, microwaves or ultrasounds. At the same time, we have developed a number of tandem transformations, such as metathesis-reductions, metathesis-oxidations, metathesis-borylations, etc. to make the chemical synthesis more efficient.
More reading
For further details, see our recent publications.


